Super Fast Ions – Solid Ion Conductor Lithium Lanthanum Titanate
What does it look like?
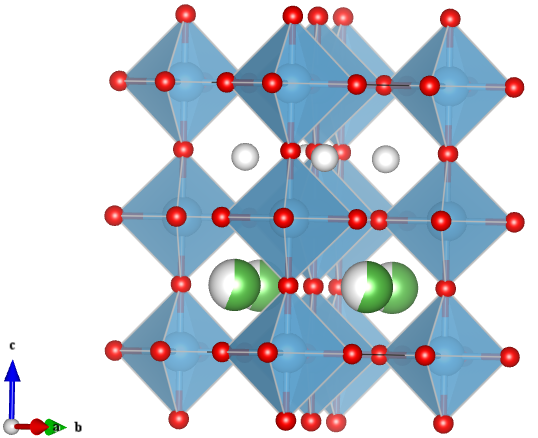
—Image generated by the VESTA (Visualisation for Electronic and STructual analysis) software http://jp-minerals.org/vesta/en/
Here the oxygen anions are red, titanium is blue, the partial lanthanum occupancy is indicated with the mixed grey/green and lithium is grey. While the structure is shown with two distinct layers of A-sites, in reality there is some disorder and mixing of Li and La between each layer.
What is it?
The Lithium lanthanum titanates, Li3xLa2/3-xTiO3 (LLT) are a series of compounds which are themselves part of a subset of perovskites known as A-site deficient perovskites. Originally discovered by Brous et al. in 1953, members of this perovskite series were originally studied for their potential dielectric properties arising from displacements of the Ti4+ cation.[1,2] However, there was not a lot of success in this area due to another physical property which lead to the discovery of LLT’s most impressive and significant application as a solid state electrolyte.
It was mentioned in an earlier post that the perovskite structure consists of a large A-site cation inside a cage created by BX6 octahdra where the B-cation is smaller than the A-site and X is an anion. In LLT, lithium and lanthanum both occupy the A-site, the B cation is Ti and X is oxygen. However, unlike standard perovskites, not every A-site is occupied for most compositions in the Li3xLa2/3-xTiO3 series, resulting in vacancies. In addition, the lithium does not actually sit in the centre of the A-site, but rather slightly off-set towards the oxygen window between A-sites. These unique structural aspects are what give rise to the extremely fast lithium ion conductivity exhibited by these materials. In fact, the specific composition Li0.34La0.51TiO3 has the highest lithium ion conductivity compared to all other solid ion conductors at room temperature. [3] Impressively, the lithium ion conductivity comes close to even that of liquid electrolytes currently used in lithium ion batteries (1.53 x 10-3 S cm-1 for LLT compared to 1 x 10-2 S cm-1 for LiPF6 dissolved in dimethylcarbonate and ethylene carbonate). Incidentally, the high ion conductivity is what makes LLT a poor dielectric material.
Where did the structure come from?
While the basic LLT structure is well known, there are many contradicting reports to the exact structure. It has been determined through a combination of variable temperature neutron, synchrotron and electron diffraction that the structure, and resulting physical properties, are dependent on the exact composition and thermal history of the compound. The most commonly reported structure – the layered tetragonal defect perovskite structure is provided below for Li0.3La0.567TiO3.
[1] Brous, J.; Fankuchen, I. & Banks, E., Acta Crystallogr., 1953, 6, 67-70
[2] Inaguma, Y.; Chen, L.; Itoh, M.; Nakamura, T.; Uchida, T.; Ikuta, H. & Wakihara, M. Solid State Commun., 1993, 86, 689-93
[3] Stramare, S.; Thangadurai, V. & Weppner, W. Chem. Mater., 2003, 15, 3974-3990