Square snowflakes – the structure of Ice VI
What does it look like?
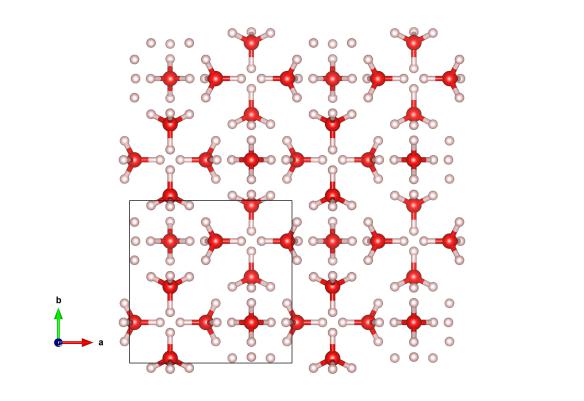
Image generated by the VESTA (Visualisation for Electronic and STructual analysis) software http://jp-minerals.org/vesta/en/
What is it?
You can freeze water by cooling it, but did you know that you can also freeze water by pressurising it? It takes quite a bit of pressure (0.9 GPa, about 9000 times atmospheric pressure or about the end of a stiletto heel with a fully grown African elephant wearing it), but at this point water will become solid at room temperature. But things are a little strange under pressure, and rather than forming the hexagonal ice, the water atoms will arrange themselves into a tetragonal structure, called Ice VI. One of the implications for this is that, rather than the six-sided hexagonal snowflakes and crystals that we are used to, water under these pressures will form square crystals!
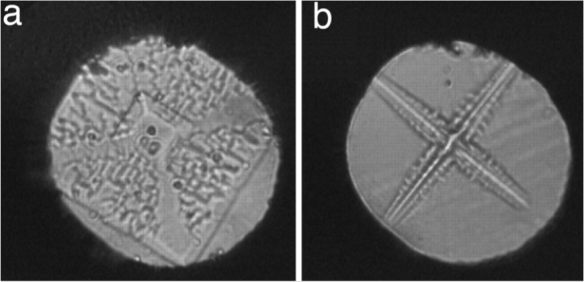
Microphotographic images of pressure-induced dendritic Ice VI crystals (a) and (b) from work by Lee et al. http://www.pnas.org/content/104/22/9178.figures-only
Where did the structure come from?
Though Ice VI was discovered by Percy Bridgman in 1912, its structure wasn't known until Kamb's work in 1965. Even then he could only postulate on the position of the water atoms (i.e. the oxygen atom positions) as the X-rays he used couldn't see the hydrogen atoms. This had to wait until scientists could make a sample large enough to use neutron diffraction in 1984, to find the positions of all the atoms.