Collagen: Triple strength
 What is it?
What is it?
The best designs – in architecture, engineering, software development – are those that effectively solve a functional problem. The same is true in biology, and scientists have long since appreciated that a protein's design is intrinsically related to the particular function that it carries out. This idea is exemplified by collagen, a highly abundant protein in the body found in the connective tissue of tendons, skin and ligaments. Collagen has a triple helical structure in which three individual chains are wound together to form a tightly organized protein rope. This organization lends collagen a high tensile strength enabling it to function as a robust structural protein that provides a scaffold within tissue and connects it to bone.
What does it look like?
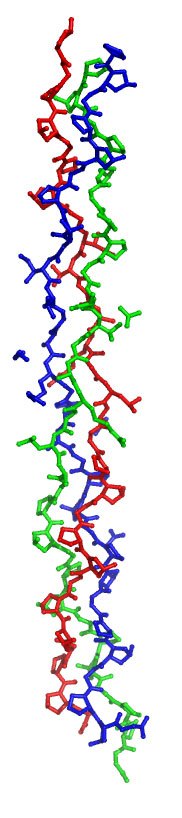
Collagen is made up of three polypeptide chains (red, green, blue) wound tightly in a triple helical structure. The individual polypeptide chains have a very distinctive and unusual sequence in which every third amino acid is a glycine (the smallest amino acid) that faces into the interior of the protein rope. This pattern allows the three chains to pack snugly against one another without clashing. Collagen also has an unusually high proline content. Proline is a rather geometrically constrained amino acid and its abundance helps the individual chains to spontaneously adopt a stable left handed helical conformation.
Where did the structure come from?
This structure is of a model peptide of Type III collagen that assembles as a homotrimer. It is a small part of the much larger protein that naturally exists in the body but it reveals the triple helical rod like structure of the protein. It was reported in Nature Structural Biology in 1999 and is PDB ID 1BVK1.
1. Kramer et al., Nature Struc. Biol 1999. 6:454-457