Getting in a Twist – A Polyrotaxane
Dave Turner tells us about some of the crystal structures he's been working on.
What does it look like?
What is it?
My imagination for finding vaguely amusing molecules, or ones with an amusing back story, has temporarily deserted me. In times of need there is only one truly reliable person to turn to. That's right, today I'm posting my own work for the first time!
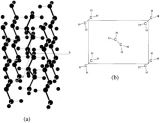
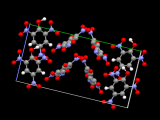
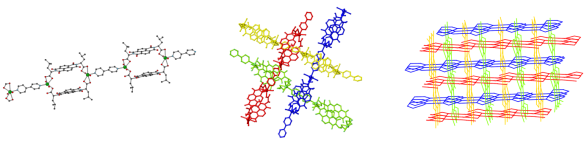
My group has recently been interested in using dicarboxylates to make polymeric structures by forming bridges between metal atoms, giving a type of material known as a coordination polymer. In this specific example, we form chains that contain rings which are bridged by another organic group, bipyridine (see picture, left). The size of the ring is ideal for a bipyridine to pass through and the chemical nature of these groups means that it is favourable for it to do so. The structure is able to satisfy this inclusion by having chains that run at angles to each other and pass through each other. The middle picture shows four chains, with the bipyridine of one passing through the ring of another. As these chains run the length of the crystal, overall we see a complicated arrangement with each chain passing through, or being threaded by, many others (see picture, right). An arrangement where a molecular thread passes through a molecular ring is known as a rotaxane, with this example being a polyrotaxane.
We make these types of structures to try and control the manner in which molecules assemble in the solid state. Unbelievably we were actually trying to get something similar to this when we carried out the reaction.
These structures always make me think about crystal formation. For crystals made from simple molecules it is easy to envisage the identical molecules packing on top of each other, like playing Tetris with only one type of block (and yes, that’s a time-wasting hyperlink for you). In these complicated structures that have rings being threaded through each other, the process is much harder to imagine and it is impressive that such arrangements can form.
Where did the structure come from?
We published this structure earlier this year, alongside related examples of interlocked chains that form polycatenated structures. The work appeared in Chemical Communications, and was the work of a final year undergraduate in my research group.