Penicillin – D-Day
June 6 – Today is the 70th Anniversary of D-Day and Crystallography 365 marks the culmination of a remarkable research project which crossed many boundaries to bring together scientists for the betterment of humankind – national, commercial and academic jealousies were suppressed in the concerted effort to develop the first antibiotic able to be produced on an industrial-scale. Prior to the production of penicillin, planning for the D-Day invasion had to take into account the potential for enormous numbers of fatalities due to inevitable infections of wounds which were in other respects survivable. The success of this endeavour led to a revolution in medicine with enormous reductions in deaths and serious illness from bacterial infections which had previously been a major cause of death.
What is it?
Penicillin is a "fused ring system" molecule with a 5-membered thiazolidine ring S-C1-C2-C3-N-C5 which shares N and C5 with the adjacent beta-lactam ring N-C5-C6-C7 which has an oxygen joined to C7 and a nitrogen joined onto C6 as below.
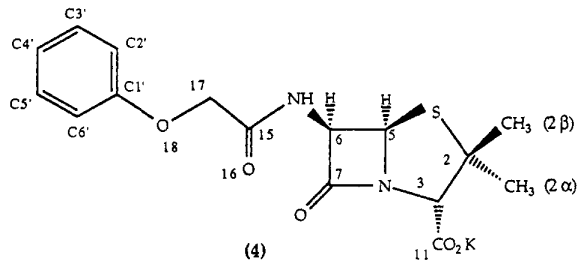
This molecule is penicillin-V and is shown as the potassium salt. The molecule produced for D-Day was penicillin-G which does not have O18. In penicillin-G C17 joins directly onto C1'.
What does it look like?
An "old-fashioned" penicillin-G model is shown in the Google doodle below (released on May 12 – the 104th Anniversary of Dorothy Hodgkin’s birth). The shape of the penicillin molecule leads to rupture of bacterial cell walls which stops the bacterium from infecting cells and causing illness.
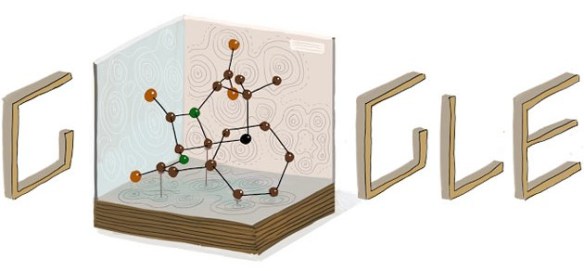
Where does it come from?
Penicillin-G was first isolated from a mould – many species of Penicillium fungi produce different types of penicillin as metabolites. Following the determination of the structure of penicillin methods were found to synthesize a range of penicillins with variations in their structure – particularly in the attachments at C17. The discovery of the Pen-V variant provided an acid stable antibiotic which could be efficiently administered orally. This variant also has superior stability on heating and could be distributed much more easily as it does not require refrigeration.
The structure of penicillin was first determined by Dorothy Hodgkin using X-ray diffraction methods as part of the huge war-time effort to develop antibiotics (follow this link to hear Dorothy speak of her collaboration with Charles Bunn at ICI to use a "fly's-eye" method to help advance the structure calculations). When John Cornforth (Nobel Prize in Chemistry 1975) who was also involved in the penicillin project first saw the proposed structure he is reported to have said "if that is the formula of penicillin I will give up chemistry and grow mushrooms!"