What does it look like?
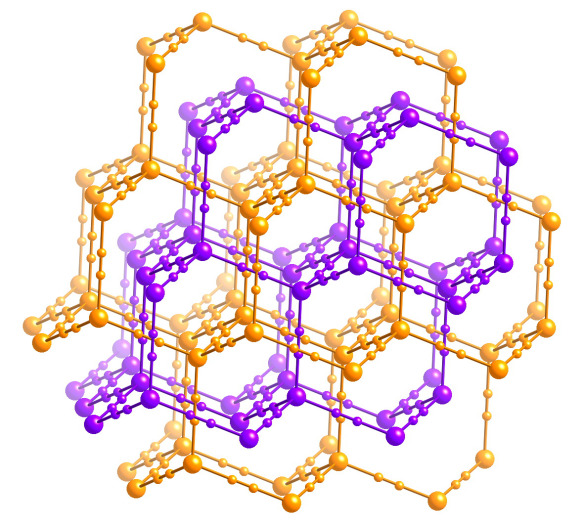
What is it?
In April 1941 a Russian scientist by the name of G. S. Zhdanov published the structure of zinc cyanide. He followed up with a second paper in 1945 describing the structure of the cadmium analogue, which is isomorphous (has the same structure).
The structures consist of infinite arrays of metal atoms bridged by cyanide anions, in an arrangement similar to the carbon atoms in diamond. A beautiful feature of these structures, however, was that they contain two completely separate three-dimensional networks that interpenetrate. That is, they passed through each other without ever actually being connected. Similar to links in a chain, except instead of being discrete loops passing through each other, they're infinite networks. This could well have been the first examples of structures with interpenetrating networks, and it must have been puzzling at first for Zhdanov.
Another remarkable thing about this work is that April 1941 was just two months before Hitler launched Operation Barbarossa (the German invasion of the Soviet Union), and by 1945 the German invasion had been repulsed, and the Russians had returned the favour by marching on Berlin. So while most of Europe was ablaze, Zhdanov was making numerous careful measurements and hundreds of hours of hand calculations, working out the crystal structure of these two materials, which would have taken months if not years. In Moscow. In the middle of World War 2. Now that's dedication.
Where did the structure come from?
"Crystalline structure of zinc cyanide", G. S. Zhdanov, Doklady Akademii Nauk SSSR, 1941, 31, 352.
"The crystal structure of cyanides. II Structure of cadmium cyanide", E. A. Shugam and G. S. Zhdanov, Acta Phys. URSS, 1945, 20, 247.