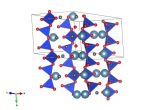
What's in silly putty? – Borax
What does it look like?
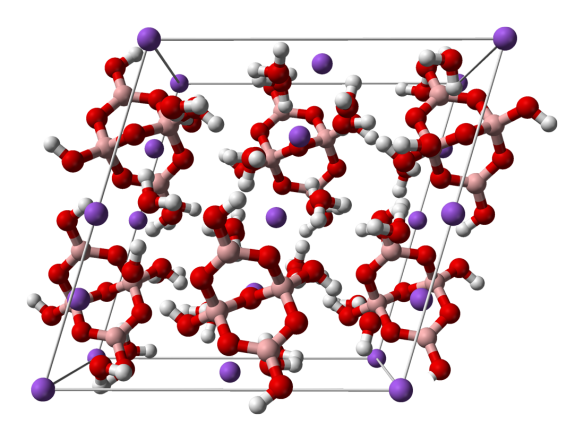
What is it?
Sodium tetraborate, more commonly known as Borax, is a substance that has been used for centuries for all sorts of purposes. First imported from Tibet via the Silk Road, it has been used as a fire retardant, an anti-fungal, and even a chemical additive in the metal industry. But today we'll just tell you about silly putty.
Mix borax with water and polyvinyl acetate (PVA) glue and you can make silly putty! The borate molecules get cross linked with the polyvinyl within the glue, to make this material strong in short time scales and flowing on a longer timescale!
Where did the structure come from?
This structure of borax (which, in fact is a hydrate as sodium tetraborate usually crystallises with water) was determined by Levy and Lesensky in 1978 at the same time that they determined the structure of Glauber's salt.