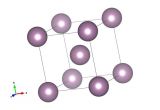
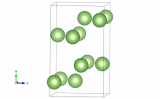
A super cool material – the crystal structure of Krypton
What does it look like?
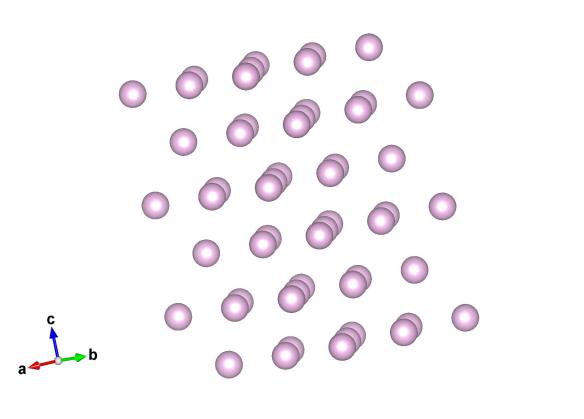
Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
The first of our requested crystal structures. Well, nearly… On Twitter we asked 'What crystal structure would you like to see blogged about' and our friends @Aus_ScienceWeek asked for Kryptonite. OK, if we had access to kryptonite (of the green variety) I’m not sure we'd be doing crystallography, we'd be finding other ways to save the world. But how about the element Krypton, that's close, right?
Krypton is a noble gas, and so not something we tend to think of as a solid crystalline material. Like the other noble gases, argon and neon to name a couple, they have full outer shells of electrons, no room to accept any others and none to give away for the formation of bonds with other elements. In fact the first compound with a noble gas wasn't discovered until 1962. It's this full shell that makes the noble gases pretty unreactive; they like to sit on their own being, well, noble. This unreactivity makes them pretty useful for creating neutral environments to play with other materials. Lots of gloveboxes will be filled with argon, and lightbulbs will often be filled with neon and other noble gas mixtures.
But what if we cool it down? Krypton will become a liquid at about -153°C but only stays liquid for a very narrow window as it will freeze to a solid at -157°C. Solid Krypton is an example of a hexagonally close packed solid, the other most efficient way of packing spheres to cubic close packing (the structure that gold takes up).
Where did the structure come from?
This krypton structure is #9012404 in the Crystallography Open Database.