An impossible molecule – Hexaferrocenylbenzene
What is it?
With the discovery of ferrocene in the 1950s, chemists realised that this was only the starting point for many other materials with properties that would be of use to us. From this point they started to build other molecules from a combination of metal atoms and organic molecules.
One molecule that was sought for a long time was hexaferrocenylbenzene – which would be made up of six ferrocene sandwiches attached to a benzene ring. The community was split as to whether such a molecule was possible, but were encouraged by its potential properties as a 'molecular gear'.
What does it look like?
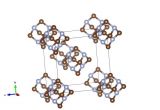
In 2006 it was finally synthesised and its crystal structure worked out – here's the unit cell. 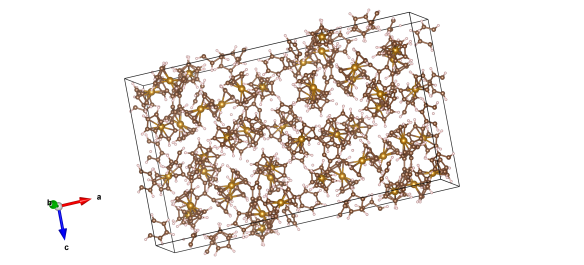
Breaking that down a bit, here is an image of the hexaferrocenylbenzene itself within the structure.

Where did the structure come from?
Hexaferrocenylbenzene was synthesised by a group at the University of California, Berkley in 2006. They subsequently worked with a group of crystallographers to determine its structure which was published in Chemical Communications. You can get the crystallographic information file for this structure directly from the supplementary information of the original paper.