Crystals, chirality and Pasteur
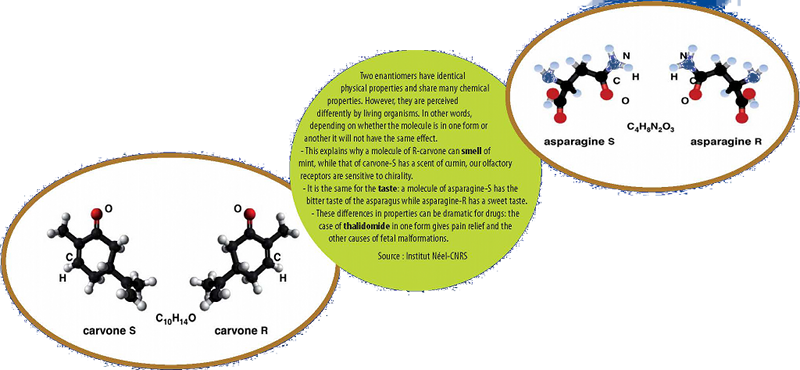
The concept of chirality is very important in many branches of science. It is responsible for properties such as the direction of rotation of plane polarized light, the taste and smell of chiral compounds, and it is fundamental to the chemistry of living organisms.
Chiral comes from the Greek word chiro which means 'hand'. When the palms are turned towards the sun, the left hand cannot be superimposed on to the right hand.

The chirality of molecules
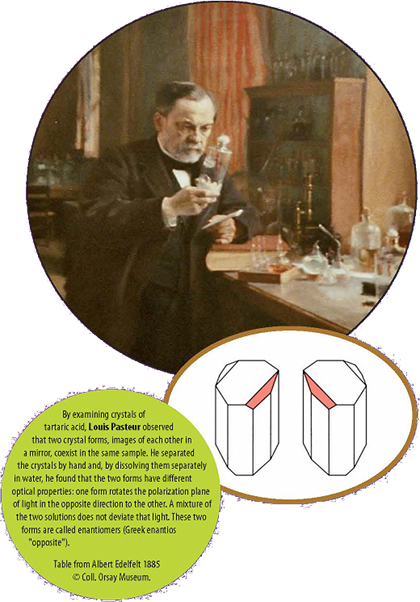
In 1848, Pasteur commented that crystals can have two identical and yet opposing forms, a mirror image of each other. He interpreted it as the existence of two chiral molecules. The chirality of crystals is primarily due to the manner in which the constituent atoms or molecules are arranged. Asymmetric molecules have two chiral forms; generally in nature one of these two forms is dominant.
Our body is made up of basic chiral structures: amino acids, sugars . . .
A chiral molecule in one isomer or another will not have the same effect on our bodies. This is the case with many drugs and medicines, as well as in the perception of tastes and smells.
A chiral molecule in one isomer or another will not have the same effect on our bodies. This is the case with many drugs and medicines, as well as in the perception of tastes and smells.