Conducting heat with a very complicated structure
What does it look like?

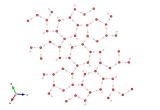
Image generated by the VESTA (Visualisation for Electronic and STructual analysis) software http://jp-minerals.org/vesta/en/
It shows a view along the <110000> direction, drawn over the equivalent of 8 × 8 × 8 fluorite-type subcells. The fractional occupancy cutoff for drawing atoms is 1/2. Bi atoms are yellow, Nb atoms and polyhedra are blue, oxygen atoms are red.
What is it?
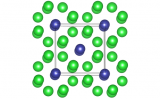
The high-temperature form of bismuth oxide, δ-Bi2O3, is the best solid-state oxide-ionic conductor known. Pure δ-Bi2O3 has never been seriously considered for practical applications (such as fuel-cell membranes) due to its limited thermal stability, but chemically stabilised variants that preserve its average structure to room temperature, along with a significant part of its conductivity, have genuine potential. The most elegant and effective of these are the Type-II phases, which show the highest conductivities across the widest ranges of chemical and thermal stability, but remain poorly understood because none of their (3+3)-dimensional "hypercubic" structures has even been determined.
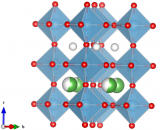
A large (cm-scale) crystal of Type-II Bi2O3–Nb2O5 was grown by refluxing bismuth oxide in a floating-zone furnace. Neutron diffraction data was collected from this crystal on the instrument D19 at the Institut Laue-Langevin (France) and solve what is by far the most complex and detailed structure of its type. The result reveals ordered strings of corner-connected NbO6 octahedra separating continuous disordered oxygen-deficient channels, explaining its substantially higher conductivity compared to other stabilised δ-Bi2O3 phases. This is the first-ever full (unconstrained) refinement of a 3+3-dimensional incommensurately modulated structure.
Where did the structure come from?
A crystallographic information file can be obtained as part of the Supporting Information associated with: Ling, C.D., Schmid, S, Blanchard, P.E.R., Petricek, V., McIntyre, G.J., Sharma, N., Maljuk, A., Yaremchenko, A.A., Kharton, V.V., Gutmann, M., Withers, R.L., J. Am. Chem. Soc. 2013, 135, 6477–6484. http://dx.doi.org/10.1021/ja3109328