LiFePO4 – The Unexpected Battery Success Story
What does it look like?
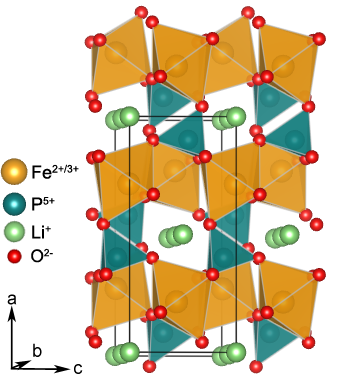
Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
Two of the basic requirements of positive electrode materials for lithium ion batteries are that they must be both good electronic and good ionic conductors. This enables the material to both accept electrons from the external circuit and insert lithium ions from the electrolyte.
LiFePO4 is not a good electronic or ionic conductor and yet it is the most promising positive electrode materials introduced in the last 10 years. Its success came as a result of a thorough understanding of the structural changes during lithium insertion and clever manipulation of the particle shapes and morphology.
The structure of LiFePO4, which is a naturally occurring mineral called triphylite, was first explored by Bjoerling et al. in 1938.1 The significance of LiFePO4 as a positive electrode material was then reported by Padhi et al.2 The LiFePO4 olivine structure is formed by a hexagonal close packed oxygen array with Li+ and Fe2+ occupying half of the octahedral sites and phosphorus occupying 1/8 of the tetrahedral sites in between the layers. Each FeO6 octahedron is corner linked to other FeO6 octahedra to form zig-zag planes running parallel to the c-axis within alternating a-c planes. In addition, one edge of the FeO6 octahedron is shared with one PO4 tetrahedron and two edges are shared with two LiO6 octahedra. The LiO6 octahedra form linear chains of edge shared octahedra between layers of FeO6 octahedra and share edges with two PO4 tetrahedra (see Figure).
The electrochemical properties of LiFePO4 are strongly influenced by its structure. For example, the inductive effect of the PO4 leads to higher voltages, enabling normally uninterested cations, such as iron, to be used successfully in a positive electrode material. But there are still problems; the PO4 is also responsible for the drastically reduced electronic conductivity in these materials. Further, the lithium diffuses through the material via one-dimensional channels which greatly inhibits the ionic conductivity.
Both of these shortcomings were overcome by preparing nanoscale LiFePO4 particles coated in a thin layer of carbon black. The increase in electronic conductivity was understood to be due to the thin carbon black layer providing a pathway for electrons from the particles to the current collector. The drastic increase in ionic conductivity, however, was not expected from a reduction in particle size.
This is because the reaction was observed to proceed via a two phase conversion reaction between LiFePO4 and FePO4. Thus, the main energy barrier to lithium extraction is the seeding and growth of the second phase. A solid solution reaction, where there is a gradual conversion from LiFePO4 to FePO4 via LixFePO4 (0<x<1), on the other hand benefits from a reduction in particle size due to shorter diffusion pathways.3 It was hypothesised, and then eventually proved, that the lithium insertion reaction proceeds via a pseudo-solid solution type reaction where the reaction front between the two phases is single phase LixFePO4.
This behaviour is highly unusual and the main reason why LiFePO4 has become a significant new battery material. So, in future, it is likely that batteries for electric vehicles as well as portable electronic devices will contain LiFePO4 as the positive electrode material.
Where did the structure come from?
LiFePO4 is #1011090 in the Crystallography Open Database.
- Bjoerling, C. O., Westgren, A. Minerals of the Varutrask pegmatite. IX. X-ray studies on triphylite, varulite, and their oxidation products. Geologiska Foereningens i Stockholm Foerhandlingar 60, 67-72 (1938).
- Padhi, A. K., Nanjundaswamy, K. S. & Goodenough, J. B. Phospho-olivines as positive-electrode materials for rechargeable lithium batteries. Journal of the Electrochemical Society 144, 1188-1194 (1997).
- Malik, R., Zhou, F. & Ceder, G. Kinetics of non-equilibrium lithium incorporation in LiFePO4. Nat. Mater. 10, 587-590 (2011).