Confining water – the structure of HHTP tetrahydrate
What does it look like?
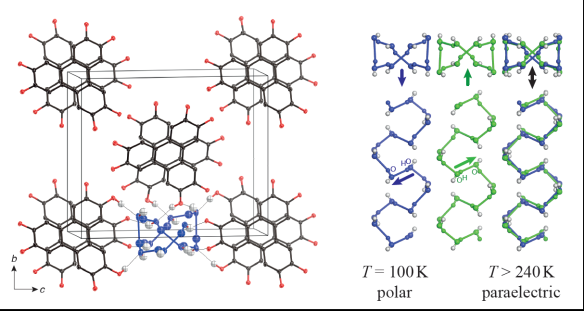
The picture was generated by CrystalMaker and MacPyMOL software.
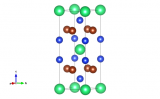
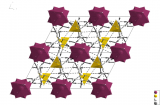
The structure of 2,3,6,7,10,11-hexahydroxytriphenylene (HHTP) tetrahydrate involves π-stacking of the disc-shaped HHTP molecules into columns, which are arranged in a pseudo-square lattice. The structure is porous and the pores between the HHTP stacks are filled with water molecules, where hydrophilic hydroxyl groups on the edge of the HHTP molecules act as chemical anchors for the water chains.
What is it?
Water is perhaps one of the most well studied substances due, in part, to its pervasiveness in the environment, the essential role it plays in biological systems and its remarkable physical properties. More solid forms of water have been documented than any other material yet, despite the best efforts of numerous researchers, the dipolar nature of the water molecule has not yet been observed unambiguously to induce ordering – creating a polar ice – when originating from a proton-disordered solid.
One approach to achieving a ferroelectric response in solid water is to place it in a confined geometry where spatial limitations mean the molecules are restricted to have a correlated, polar arrangement. The use of confined water chains has near-accomplished this where ferroelectric ordering has indeed been observed but only on cooling from a liquid state. Clearly, if ice is to be exploited in switchable devices, solid-solid transformations will be far more desirable.
The present structure undergoes a polar to non-polar phase transition with increasing temperature. At low temperatures, the HHTP molecules mediate weak interactions (ca. 0.9 kJ mol-1) between the water chains and lead to the generation of an overall dipole moment with the dipole moments of each water channel pointing in the same direction. On raising the temperature above a modest 240 K, sufficient energy is supplied to disrupt the inter-chain communication, causing the system to adopt a paraelectric state. It is noteworthy that the phase transition from a polar to a non-polar structure solely originates from alignment of the water molecules. What remains now is to measure the strength of the ferroelectric response in the ordered, low-temperature phase.
Where did this structure come from?
The structures are deposited in the Cambridge Crystallographic Data Centre; CCDC 962532 for the high temperature structure and CCDC 962533 – low temperature structure.
The variable temperature study is published in Physical Chemistry Chemical Physics in 2014.