The two-coloured stone – Iolite
What does it looks like?
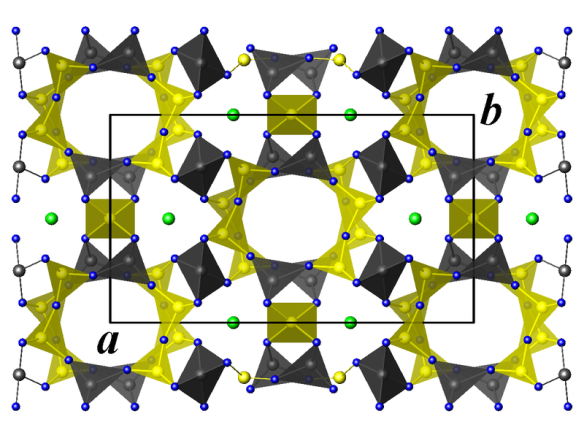
Crystal structure of cordierite. Green: magnesium atoms, blue: oxygen atoms, yellow: silicon and aluminium atoms.
What is it?
Yesterday, we blogged about labradorite and its pretty play on colours. Today we're introducing cordierite, otherwise known in gem-quality form as iolite, the two-coloured 'stone' (Greek ἴον, violet + λίθος, stone). It's another of the beryl family, along with Emerald and Aquamarine, but the ratio of iron and magnesium in this structure means that it can change colour. Known as pleochroism, this effect occurs depending on the angle of the light shining on it. Iolites can change from a deep blue to a watery grey.

Pleochroism of Cordierite – Left image same specimen in polarized light. Image by Didier Descouens.
It is this feature that earned this material the nickname 'water sapphires' or 'Viking compasses'. The latter name probably stems from the thought that the Vikings used these gems to find the direction of the sun on a cloudy day (as it would affect the colour of the crystal).
Where did the structure come from?
Entry #9000998 in the Crystallography Open Database is a study on how sodium affects the cordierite crystal structure.