October birthstone: Opal. When is a crystal not quite a crystal?
Helen Brand continues her series on the crystal structures of birthstones:
This month's birthstone is opal. On the face of it I haven't been looking forward to this post because opal is an "amorphous" material, so finding an angle to write about in a blog dedicated to crystal structure was going to be tricky. Luckily, it is still sort of crystal-y … Opal is a mineraloid – mineral-like material. Chemically it is a hydrated form of Silica, SiO2, which has been talked about several times already on this blog and is a building block for our planet. Opal is a low-temperature deposit of silica which is typically found in fissures in rocks.
The story of the structure of Opal is a largely Australian one. Figure 1 shows a fire opal from Andamooka, South Australia. Opals are the national gemstone of Australia and it is estimated that approximately 95–97% of all opals are found in Australia. The map below (Figure 2), shows where the major opal deposits are in Australia. The first opals were boulder opals which were found in Queensland in the 1870s.

Figure 1. A fire opal from http://www.minerals.dmitre.sa.gov.au/
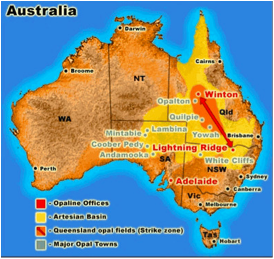
Figure 2. Opal deposits in Australia from http://www.opalauctions.com
The variety of internal colours which mark the opal is a result of the internal structure of the opal. Hang on, isn't it amorphous? Yes and no. An opal is composed of a series of "amorphous" silica spheres. These spheres are then arranged in a regular array – a close-packed lattice. Just like in a crystalline solid – in this case it is particles which are packing rather than the atoms.
This means that the length-scales of the repeating array are different from a traditional crystal. They are of the order of a few hundred nanometers rather than tens of nanometers. Thus, to probe the structure using diffraction, a radiation has to be chosen where the wavelength is similar to the distance between the layers of silica spheres – a few hundred nanometers. On the electromagnetic spectrum, the energy which corresponds to these wavelengths is visible light (see Figure 3). This means that when we see colour in an opal, we are actually "seeing" diffraction from the layers of silica spheres.
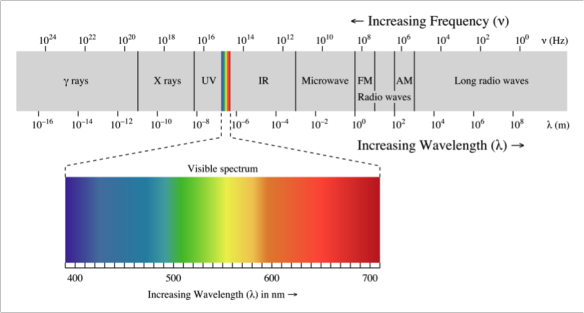
Figure 3. Electromagnetic spectrum. http://www.wikipedia.com
In the 1960s, J. V. Sanders was investigating Opals at CSIRO in Melbourne. He used an SEM to image opals and discovered the regular array of silica spheres. Figure 4 below shows two of the SEM images he obtained from the surface of one of the opals. Sanders then went on to analyse the structure further using light diffraction to determine the nature of the packing. He determined that there are layers which are arranged hexagonally and can be packed either as a face-centred cubic sequence or a hexagonal close packed sequence. Figure 5 shows an example of the light diffraction spots he obtained from an opal using a mercury–cadmium lamp.
If we think about it, we can use the colour of the opal to tell us a rough size of the silica spheres which make up that particular opal. Looking at Figure 3 – an opal with an average of 400 nm between the layers of spheres will show indigo and blue colours while an opal with an average of nearer 700 nm between the layers (thus larger spheres), will show more red and orange colours.

Figure 4. The Surface of an opal. From Sanders (2014). Colour of precious opals. Nature 204, 1151–1153.
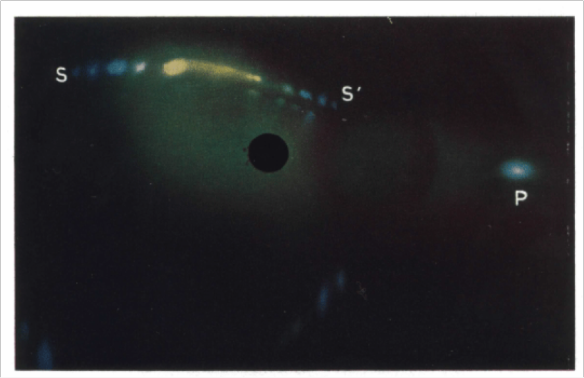
Figure 5. Light diffraction image of an opal obtained using a mercury–cadmium spectral lamp. From: Sanders (1968). Acta Cryst. A, 24, 427–434.
References:
- V. Sanders (1964). Colour of precious opals. Nature, 204, 1151–1153.
- V. Sanders (1968). Diffraction of Light by Opals Acta Cryst. A, 24, 427–434.