Beautifully holding buildings together – Tricalcium aluminate
What is it?
Hello, it's Helen here. As we move into the final furlongs of the crystallography 365 blog we're scrambling to find structures that we've missed and really deserve a post. I know that you're not meant to have a favourite in the over a million crystal structures that have been discovered – but between you and me, this is mine. Who else would love a mineral found in cement?
Cement, when you look into it, is a very sophisticated material. It's a powder, that when you add water to can then be poured – but hours later it's tough enough to build skyscrapers from. How it does this is due to the interplay of all the mineral phases in that powder – one important one is Ca3Al2O6 or tricalcium aluminate.
Tricalcium aluminate has a bit of a love/hate relationship with cement. It's responsible for many of the impurities in the powder, but is needed because of its amazing abilty to grab water and help make the right silicate phases that give set cement its strength.
So, don't keep us in suspense… what does it look like?
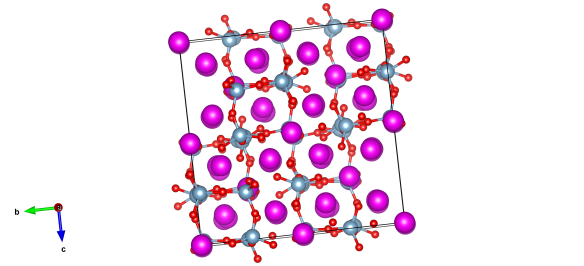
You see I love this structure because it's so complex! The interplay of forces between the aluminate (the light blue and red atom units) and the calcium atoms (in hot pink) are just enough to shift them apart so they don't line up. This means that the repeating 'unit cell' structure has to be extraordinarily big to describe them all.
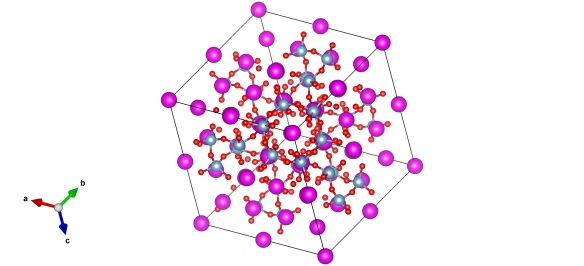
Though this is tricky to measure and describe, this 'kinked' structure is why this material reacts so strongly with water. A further upside is it does look rather beautiful.
So tricalcium aluminate – builds skyscrapers and has an incredible crystal structure – what's not to love?
Where did the structure come from?
This structure of tricalcium aluminate was found by Mondal and Jeffery in 1975, and is #1000039 in the Crystallography Open Database.