Learn about crystallography through watching
Below are listed some interesting video clips, webcasts, television programmes and films that explain crystallography. Click on the large images to download a video file, watch the clip, or be directed to an external website. Click on the smaller images for more information.
A selection of videos from the International Year of Crystallography playlist on YouTube
Diving into the heart of the molecules of life
Taking atomic snapshots is very important to biologists who want to understand the architecture of the macromolecules that make up our cells. However, these nano-objects are far too small to be observed with a microscope. This is why other methods such as X-ray crystallography are needed to provide accurate images at an atomic scale. This film, Diving into the heart of the molecules of life, follows the different steps of a crystallographic study and describes the work of researchers at CNRS (ARN Laboratory, Institut de Biologie Moléculaire et Cellulaire, Strasbourg), from the purification of a macromolecule from living cells, its crystallisation and the analysis of the crystals using synchrotron radiation, to the assembly of a three-dimensional image. This movie covers several aspects of scientists' work which are not widely known: the cloning of biomolecules, the use of large instruments such as synchrotron X-ray sources, and 3D visualisation.
Category: Education. Duration: 13m 26s
The Royal Institution Crystallography Collection
Understanding crystallography: Part One
From protein to crystal
How can you determine the structure of a complex molecule from a single crystal?Professor Elspeth Garman take us on a journey into the world of crystallography - from protein production and purification to growing the right type of crystals.
In her laboratory at the University of Oxford Elspeth introduces us to some of the tricks of the trade, key techniques and the machines that help her team grow crystals ready for X-ray analysis at specialist facilities like Diamond Light Source.
This film was supported by the Science and Technologies Facilities Council (STFC).
Published: 2014
Filmed: 2013. Duration: 7m 45s
Credits: Ed Prosser / Royal Institution
The Mystery of the Giant Crystals
![[Inside the Naica cave]](https://www.iycr2014.org/__data/assets/image/0006/102849/Poster-del-Misterio-en-ingles.jpg) The film El Misterio de los Cristales Gigantes (The Mystery of the Giant Crystals) has been made freely available by Madrid Scientific Films and Triana Sci & Tech with the support of the International Union of Crystallography as an educational contribution to the International Year of Crystallography 2014. Written and presented by Juan Manuel García Ruiz and directed by Javier Trueba, the film tells the story of the scientific investigation into the nature and properties of the giant gypsum crystals found in a silver mine in Mexico in 2000.
The film El Misterio de los Cristales Gigantes (The Mystery of the Giant Crystals) has been made freely available by Madrid Scientific Films and Triana Sci & Tech with the support of the International Union of Crystallography as an educational contribution to the International Year of Crystallography 2014. Written and presented by Juan Manuel García Ruiz and directed by Javier Trueba, the film tells the story of the scientific investigation into the nature and properties of the giant gypsum crystals found in a silver mine in Mexico in 2000.
Trailer |
Follow this link to read more about the project or to donate to the work of Triana Sci & Tech |
Streaming HD video
Click on the images below to view the film in high definition in English, Spanish, Italian or French.
The Mystery of the Giant Crystals
The Cave of the Crystals (Cueva de los Cristales) in the Naica Mine, Chihuahua, Mexico, houses some of the largest natural crystals ever found. They are selenite, a form of the mineral gypsum (CaSO4.2H2O). Juan Manuel Garcíia Ruiz and his colleagues investigate the conditions under which these huge crystals have grown over the course of thousands of years. The temperatures in the subterranean caverns are over 50°C, and the caves are filled with water containing a variety of minerals leached from the surrounding rocks.
Duration: 50m 53s
© 2014 Trianatech.com - All rights reserved
El Misterio de los Cristales Gigantes
La Cueva de los Cristales en la mina de Naica, Chihuahua, México, alberga algunos de los más grandes cristales naturales que se han encontrado. Son selenito, una forma del yeso mineral (CaSO4.2H2O). Juan Manuel García Ruiz y sus colegas investigan las condiciones en que estos enormes cristales han crecido a lo largo de miles de años. Las temperaturas en las cavernas subterráneas son más de 50° C, y las cuevas están llenas de agua que contiene una variedad de minerales lixiviados de las rocas circundantes.
Duration: 50m 54s
© 2014 Trianatech.com - All rights reserved
Il Mistero dei Cristalli Giganti
La Grotta dei Cristalli (Cueva de los Cristales) nella miniera di Naica, Chihuahua, Messico, ospita alcuni dei più grandi cristalli naturali mai trovati. Si tratta di cristalli di selenite, una particolare forma di gesso (CaSO4.2H2O). Juan Manuel García Ruiz e i suoi colleghi indagano sulle condizioni in cui questi enormi cristalli sono cresciuti nel corso di migliaia di anni. La temperatura nelle caverne sotterranee è oltre 50° C, e le grotte sono riempite con acqua contenente una varietà di minerali lisciviati dalle rocce circostanti.
Duration: 50m 58s
© 2014 Trianatech.com - All rights reserved
Le Mystère des Cristaux Géants
La Grotte des Cristaux (Cueva de los Cristales) dans la mine de Naica, Chihuahua, Mexique, abrite quelques-uns des plus grands cristaux naturels jamais trouvés. Ils sont sélénite, une forme de gypse minéral (CaSO4.2H2O). Juan Manuel García Ruiz et ses collègues enquêtent sur les conditions dans lesquelles ces énormes cristaux ont grandi au cours de milliers d'années. Les températures dans les cavernes souterraines sont plus de 50° C, et les grottes sont remplis avec de l'eau contenant une variété de minéraux lessivés des roches environnantes.
Duration: 50m 58s
© 2014 Trianatech.com - All rights reserved
Educational videos
Plongée au cœur des molécules du vivant
"Voir pour mieux comprendre" pourrait être la devise du biologiste qui s’intéresse à l’architecture de macromolécules composant nos cellules. Or ces nano-objects sont de très petite taille et ne peuvent être observés directement au microscope. Aussi, pour obtenir une image précise à l’échelle de l’atome, il est nécessaire de faire appel à d’autres approches comme la cristallographie aux rayons X. Cette plongée au coeur des molécules du vivant retrace les étapes d’une étude cristallographique conduisant à la visualisation d’une macromolécule biologique. Le travail des chercheurs d’un laboratoire du CNRS (ARN, IBMC, Strasbourg) est suivi pas à pas depuis l’isolement et la purification d’une macromolécule à partir de cellules vivantes jusqu’à l’établissement de son image tridimensionnelle, en passant par sa cristallisation et l’analyse des cristaux à l’aide du rayonnement synchrotron. Ce film illustre des aspects méconnus du quotidien des scientifiques comme le clonage de molécules, l’utilisation de grands instruments tels que les accélérateurs de particules, ou la visualisation 3D.
Category: Education. Duration: 13m 26s
Historical videos
Crystallographers in Conference 1965
During the 1965 International Conference on Crystallography held in Melbourne, the opportunity was taken to record some of the great figures in the history of this subject. The twin topics forming the discussion basis for the conference were electron diffraction and the nature of defects in crystals. Among the distinguished scientists present, many had made special contributions to the research on those topics - had, in fact, pioneered a place for themselves in the story of crystallography.
Filmed at the International Crystallography Conference, 1965, Melbourne, Australia, by the CSIRO Film Unit in collaboration with the Australian Academy of Science. With support from UNESCO, the International Council of Scientific Unions, the Australian Academy of Science, the International Union of Crystallography, the International Union of Pure and Applied Physics, the International Atomic Energy Agency and the University of Melbourne.
Category: Science & Technology. Duration (complete programme): 41m 44s
Licence: Standard YouTube Licence



![[Electron density map, protein model and crystal]](https://www.iycr2014.org/__data/assets/image/0009/100152/molecules_of_life.jpg)
 What crystallography can do for you
What crystallography can do for you The importance of X-ray crystallography in the materials and biological sciences
The importance of X-ray crystallography in the materials and biological sciences Growing crystals: the Canberra competition
Growing crystals: the Canberra competition The importance of crystallography in our daily lives
The importance of crystallography in our daily lives The role of crystals in human life
The role of crystals in human life UN Secretary General launches IYCr2014
UN Secretary General launches IYCr2014 IYCr Latin American Summit meeting on Biological Crystallography and Complementary Methods
IYCr Latin American Summit meeting on Biological Crystallography and Complementary Methods Final of the 2014 Crystallization Competition for Schools in Spain
Final of the 2014 Crystallization Competition for Schools in Spain ¿Qué es un cristal?
¿Qué es un cristal? Rigaku International Year of Crystallography 2014 Sponsor Video
Rigaku International Year of Crystallography 2014 Sponsor Video IUCr President introduces the International Year of Crystallography 2014
IUCr President introduces the International Year of Crystallography 2014 About the International Year of Crystallography
About the International Year of Crystallography UNESCO Secretary-General introduces IYCr2014 (English)
UNESCO Secretary-General introduces IYCr2014 (English) Cristalografía de rayos X, una historia de cien años
Cristalografía de rayos X, una historia de cien años The role of crystals in human life
The role of crystals in human life BBC World News: Crystallography: 19 December 2013
BBC World News: Crystallography: 19 December 2013 Año Internacional de la cristalografía en América Latina y el Caribe
Año Internacional de la cristalografía en América Latina y el Caribe How to grow a single crystal with Johanna
How to grow a single crystal with Johanna The fascinating world of crystallography - with Johanna
The fascinating world of crystallography - with Johanna Commemorative Postage Stamp release
Commemorative Postage Stamp release Concurso de Cristalización en la Escuela: Final 2014
Concurso de Cristalización en la Escuela: Final 2014 The role of crystals in human life
The role of crystals in human life![[celebrating crystallography displays a laser diffraction pattern]](https://www.iycr2014.org/__data/assets/image/0003/96825/understanding1.jpg)
 Sir Lawrence Bragg: crystals and gems
Sir Lawrence Bragg: crystals and gems Célébrons la cristallographie! (French language version)
Célébrons la cristallographie! (French language version) Forbidden crystal symmetry in mathematics and architecture
Forbidden crystal symmetry in mathematics and architecture The humble Braggs and X-ray crystallography: Solving the patterns of matter
The humble Braggs and X-ray crystallography: Solving the patterns of matter Seeing things in a different light: Friday Evening Discourse with Stephen Curry
Seeing things in a different light: Friday Evening Discourse with Stephen Curry Tales from the prep room: diffraction
Tales from the prep room: diffraction Mars diffracts! An epic journey into the role of X-ray diffraction in space
Mars diffracts! An epic journey into the role of X-ray diffraction in space Understanding Crystallography: Part Two. From Crystal to Diamond
Understanding Crystallography: Part Two. From Crystal to Diamond Celebrating crystallography: 100 years of crystallography
Celebrating crystallography: 100 years of crystallography Structure and Order. A century of symmetry with Judith Howard
Structure and Order. A century of symmetry with Judith Howard![[Inside the Naica caves]](https://www.iycr2014.org/__data/assets/image/0003/102846/giant_crystals.jpg)
![[Inside the Naica caves]](https://www.iycr2014.org/__data/assets/image/0020/102881/giant_crystals2.jpg)
![[Inside the Naica caves]](https://www.iycr2014.org/__data/assets/image/0003/102882/giant_crystals3.jpg)
![[Inside the Naica caves]](https://www.iycr2014.org/__data/assets/image/0004/102883/giant_crystals4.jpg)
 Georgina Ferry on X-ray crystallography
Georgina Ferry on X-ray crystallography A Case of Crystal Clarity: an adventure in X-ray crystallography
A Case of Crystal Clarity: an adventure in X-ray crystallography 5th Moroccan School of Crystallography (French or English)
5th Moroccan School of Crystallography (French or English) Crystallography and the reciprocal space (English)
Crystallography and the reciprocal space (English) Crystals - how and why to study them (Russian)
Crystals - how and why to study them (Russian)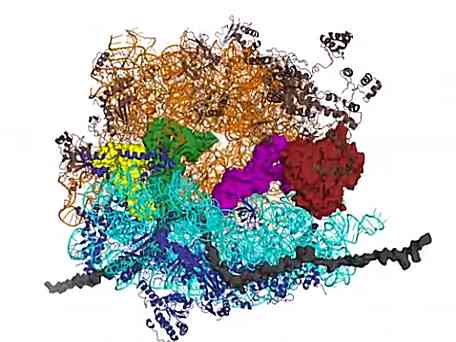 The decoding step of protein synthesis: EF-Tu delivers aminoacyl-tRNA to the ribosome
The decoding step of protein synthesis: EF-Tu delivers aminoacyl-tRNA to the ribosome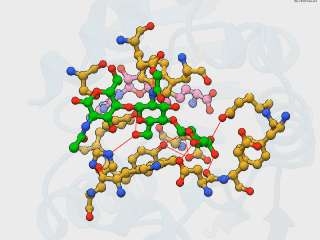 Celebrating the International Year of Crystallography with methylation of Nod Factor
Celebrating the International Year of Crystallography with methylation of Nod Factor Proyecto Krystalla: school crystallization contest in Andalusia
Proyecto Krystalla: school crystallization contest in Andalusia Revolutionary Biology Part 1: the building blocks of life
Revolutionary Biology Part 1: the building blocks of life Manchester and Bragg
Manchester and Bragg![[Crystallographers in Conference]](https://www.iycr2014.org/__data/assets/image/0011/96896/melbourne.jpg)
 Nobel winners honoured in Adelaide
Nobel winners honoured in Adelaide Dorothy Hodgkin, Biochemist
Dorothy Hodgkin, Biochemist The Braggs' legacy
The Braggs' legacy